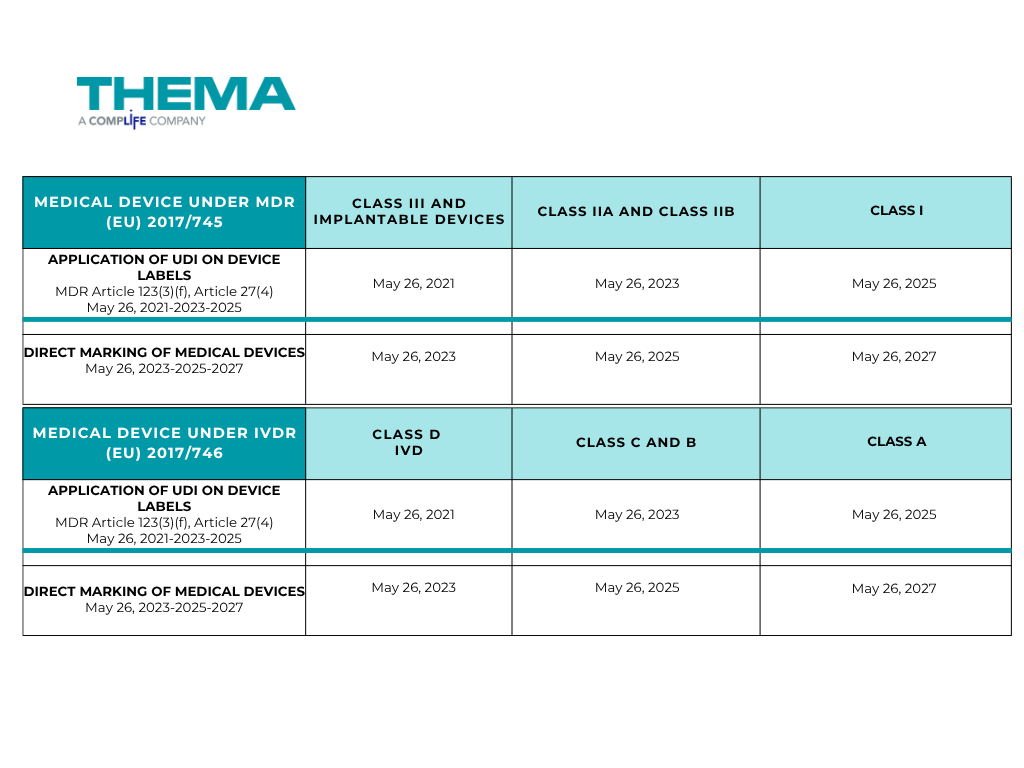
The timing of UDI implementation varies depending on the risk class of the device, as set out in the MDR (EU) Regulations 2017/745 for Medical Devices and IVDR (EU) 2017/746 for in vitro diagnostic Medical Devices.
The application dates are as follows:
For Medical Devices (MDR (EU) 2017/745):
- Applying UDI on device labels:
- For Class III and implantable devices, the deadline was May 26, 2021.
- For Class IIa and Class IIb: the deadline was May 26, 2023.
- For Class I: the deadline is May 26, 2025.
- Direct Marking of Medical Devices
- For Class III and implantable devices, the deadline was May 26, 2023.
- For Class IIa and Class IIb: the deadline is May 26, 2025.
- For Class I: the deadline is May 26, 2027.
For In Vitro Diagnostic Medical Devices (IVDR (EU) 2017/746):
- Applying UDI on device labels:
- For Class D IVD: the deadline was May 26, 2021.
- For Class C and Class B: the deadline was May 26, 2023.
- For Class A: the deadline is May 26, 2025.
- Direct Marking of Medical Devices
- For Class D IVD: the deadline was May 26, 2023.
- For Class C and Class B: the deadline is May 26, 2025.
- For Class A: the deadline is May 26, 2027.